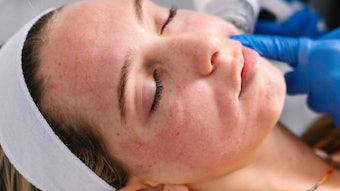
Biotechnology company Signum Dermalogix has entered into an agreement with Dermata Therapeutics, a specialty pharmaceutical company based in San Diego, to develop SIG990, an isoprenylcysteine (IPC) analog that modulates toll-like receptor (TLR) and G-protein signaling, for the treatment of patients with rosacea.
The topical anti-inflammatory compound has the potential to reduce erythema, and decrease the papules and pustules associated with rosacea. Its Investigational New Drug (IND) application for has been cleared by the U.S. Food and Drug Administration (FDA), and SIG990 is now ready to be evaluated in subjects. Dermata has obtained an exclusive worldwide license to SIG990 and is responsible for its clinical development.
“We are excited to partner with Dermata and look forward to working with their experienced team to advance SIG990 in the clinic,” said Maxwell Stock, CEO of Signum Dermalogix. “Partnering allows us to share expertise and thereby provides an effective way of progressing cutting edge research and development. I'd also like to thank the National Institute of Allergy and Infectious Diseases (NIAID) for their support in helping fund this drug development program.”