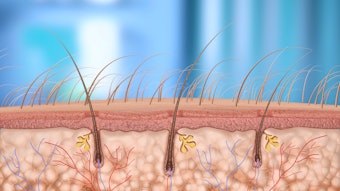
The U.S. Food and Drug Administration (FDA) has cleared Miramar Labs’ miraDry System for the treatment of unwanted underarm hair and permanent reduction of underarm hair of all colors. The miraDry System uses proprietary miraWave technology, which delivers precisely controlled microwave energy to reduce underarm hair. With this clearance, the company has announced the introduction of the miraSmooth treatment, an alternative to laser hair removal that can be offered by aesthetic providers. MiraSmooth will be available in select markets in August 2015.
“We believe the potential for microwave energy in aesthetics is exceptional,” said Michael Kleine, CEO and president of Miramar Labs. “Miramar was the first company to receive clearance for axillary sweat reduction and we are now the only company with FDA clearance for permanent reduction of axillary hair of all colors. These are both tremendous market making opportunities in aesthetics.”
Photo copyright Getty Images.