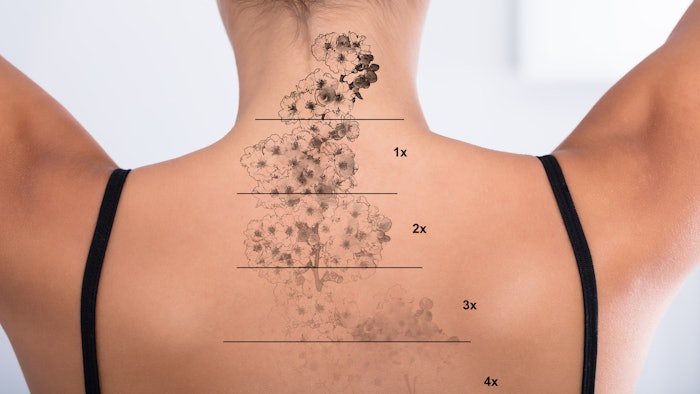
TOOsonix A/S has released its System ONE-M as a CE-marked medical device for aesthetic treatments, including those for solar lentigines, spider veins, angiomas and telangiectasia, as well as color-independent tattoo removal.
The release coincides with the publication of two clinical papers in the peer-reviewed scientific journal, Skin Research and Technology (John Wiley and Sons, Inc.), describing very positive results from the first clinical treatments performed at the Bispebjerg University Hospital, Copenhagen, Denmark by the renowned Professor Jørgen Serup, MD.
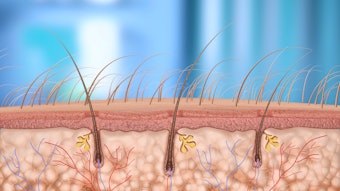
As a focused ultrasound device, the system delivers accurate thermal focal points to a chosen layer of the human skin, and directly in the lesion chosen for treatment. Applied to the outer skin the method is ablative. The system can however also reach lesions down to the dermal interface to the fat and abnormal vessels in the outer subcutis, without disturbing the out-of-focus outer skin. Thus, no wound and no scar.
“The CE marking of our first medical device is pioneering the field of HIFU used for the human skin,” Torsten Bove and Tomasz Zawada, co-founders of TOOsonix, said. “It is the first important step in TOOsonix' vision to make new and advanced HIFU systems available for dermatological use all over the globe. The new system is ready for a range of new applications. We initially intend to offer this device to first-mover dermatologists within the European Union, and look forward to the resulting physician feedback and market data that can guide our addition of more medical indications in the future.”
Professor Jørgen Serup MD, first author of the two publications introducing the system], states:
“High frequency HIFU has a significant potential for future use in dermatological clinics, particularly in the laser/IPL clinics, as a new and specialized member of the family of advanced devices. Tattoo removal has been studied in detail. In our experimental work we have furthermore, demonstrated that the device used for Actinic Keratosis has important advantages over photodynamic therapy (PDT), practically, resource-wise, and with respect to efficacy. Noteworthy is the reduced pain-level during treatment. HIFU can be applied to multiple sites of the body in one short session and, obviously, has the potential to replace PDT in the future. Experimental use indicates the device can be used for dedicated ablative treatment of skin cancers, particularly basal cell carcinoma, and for a range of different premalignant conditions as well as a multitude of benign skin tumors. The method is an entirely new treatment modality in dermatology, with a large potential.”