The National Institute of Health (NIH) has recently conducted a study with an experimental treatment for eczema, which was aimed to modify the skin microbiome safely with reduced disease severity for children as young as 3 years.
Related: Bacteria Therapy Used to Treat Eczema
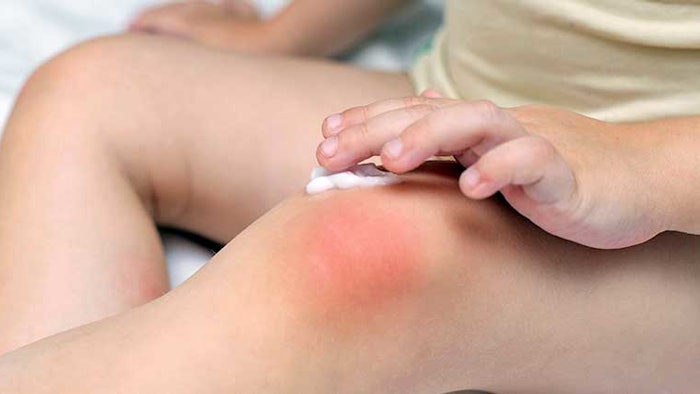
The experimental therapy contained strains of live Roseomonas mucosa, a bacterium naturally present on the skin, which was originally isolated from healthy volunteers and grown under carefully controlled laboratory conditions. For four months, clinical trial participants or their caregivers periodically applied this probiotic therapy to areas that were affected by eczema.
Numerous genetic and environmental factors contribute to eczema and scientists are learning more about the role that the skin’s microbiome plays in this condition. In 2016, NIAID researchers reported that R. mucosa strains isolated from healthy human skin improved outcomes in cell culture and mouse models of eczema.
“A child suffering from eczema, which can be itchy, painful and distracting for the child, also is very difficult for the entire family,” said study lead, Anthony S. Fauci, M.D., director of NIH’s National Institute of Allergy and Infectious Diseases (NIAID). “These early-stage findings suggest that R. mucosa therapy may help relieve some children of both the burden of eczema symptoms and the need for daily treatment.”
To build on these preclinical findings, NIAID launched a Phase 1/2 clinical trial to assess the safety and potential benefit of R. mucosa therapy in people with eczema. Interim results reported in 2018 for 10 adults and five children aged 9-14 years indicated that the treatment was deemed safe and associated to reduce eczema severity. Since then, the trial has additionally enrolled 15 children with a total of 20 children in the study with mild to severe eczema ranging from 3-16 years.
Previously: Electric Effects: EI to Measure Skin Barrier Defects
Twice weekly for three months and every other day for an additional month, children or their caregivers sprayed a solution of sugar water containing live R. mucosa onto areas of skin with eczema. For the first 15 children enrolled in the study, the dose of live R. mucosa was gradually increased each month. The last five children to enroll received the same dose throughout the four-month treatment period. Regardless of the dosing strategy, no serious adverse events were attributed to the therapy.
“Most children in the study experienced substantial improvements in their skin and overall wellbeing following R. mucosa therapy,” NIAID’s Ian Myles, M.D., principal investigator of the trial. “Encouragingly, the therapeutic bacteria stayed on the skin and continued to provide benefit after therapy stopped. These results support a larger study to further assess the safety and effectiveness of this experimental treatment by comparing it with a placebo.”
Seventeen of the 20 children experienced more than a 50% improvement from eczema severity following treatment. Improvement occurred on all treated skin sites including the inner elbows, inner knees, hands and neck. The scientists also observed increases in the skin’s barrier function and its ability to seal in moisture and keep out allergens. Additionally, most children needed fewer corticosteroids to manage their eczema, experienced less itching and reported a better quality of life following the therapy. These benefits persisted after treatment ended and the therapeutic R. mucosa strains remained on the skin for up to eight months.
Also related: Study Sheds LIGHT on Eczema Cause
The NIAID researchers then set out to better understand how R. mucosa therapy improves eczema symptoms. It was found that treated skin had increased microbial diversity and reduced levels of Staphylococcus aureus, a bacterium known to exacerbate eczema.
In addition to imbalances in the microbiome, eczema skin is deficient in certain lipids or oils. By conducting experiments in cell and animal models of eczema, the NIAID scientists found that a specific set of lipids produced by R. mucosa strains isolated from healthy skin can induce skin repair processes and promote the turnover of skin tissue. Study participants had shown increased levels of these lipids on the skin after treatment with R. mucosa.
The researchers emphasize that additional studies are needed to further elucidate the mechanism of R. mucosa therapy and to explore whether genetic or other factors may explain why some participants did not benefit from the experimental treatment.











