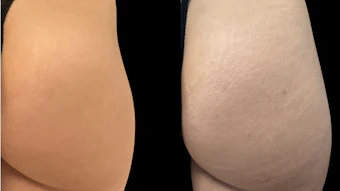
The U.S. Food and Drug Administration (FDA) has cleared Candela Corporation’s Vbeam Prima, an advanced 595nm pulsed dye laser (PDL) with an additional 1,064nm wavelength, for marketing in the United States. In addition to the added 1,064nm wavelength, the VBeam Prima offers a number of other new features to more effectively treat a broad range of skin conditions, including rosacea, port wine stains, acne, facial, leg and spider veins, scars, benign pigmented lesions and wrinkles, warts, stretch marks and photo aging.
“I’ve worked with the Vbeam platform for years, and I am extremely excited about advancements with the Prima, especially the 50% greater power enabling a 15mm spot size, both contact and spray cooling, once-a-day calibration and extended dye life thanks to a completely re-designed system,” says Eric Bernstein, MD, M.S.E., director of laser surgery & cosmetic dermatology at the Main Line Center for Laser Surgery in Ardmore, Pennsylvania. “In a clinical study using Vbeam Prima, facial redness and rosacea symptoms were dramatically reduced after four treatments.”
For more information, visit https://syneron-candela.com/na/prima