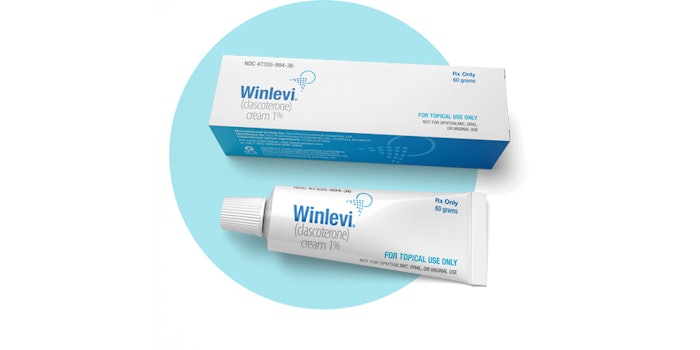
Sun Pharmaceutical Industries (Sun Pharma) has announced the availability of its WINLEVI (clascoterone) cream 1% in the U.S.
WINLEVI was approved by the FDA in August 2020 for the topical treatment of acne vulgaris in patients 12 years of age and older.
It is a first-in-class topical androgen receptor inhibitor that works by inhibiting the effects of androgen receptors in cells of the sebaceous glands to help reduce sebum production and inflammation. It is suitable for use in both males and females.
"The U.S. launch of WINLEVI enhances Sun Pharma's specialty product portfolio and reflects our commitment to meeting patients' needs by providing innovative dermatology medicines," said Abhay Gandhi, CEO of Sun Pharma North America. "With its safety and tolerability profile, combined with its demonstrated efficacy in clinical trials, WINLEVI has the potential to be an important topical treatment option for the millions of Americans affected by acne vulgaris."