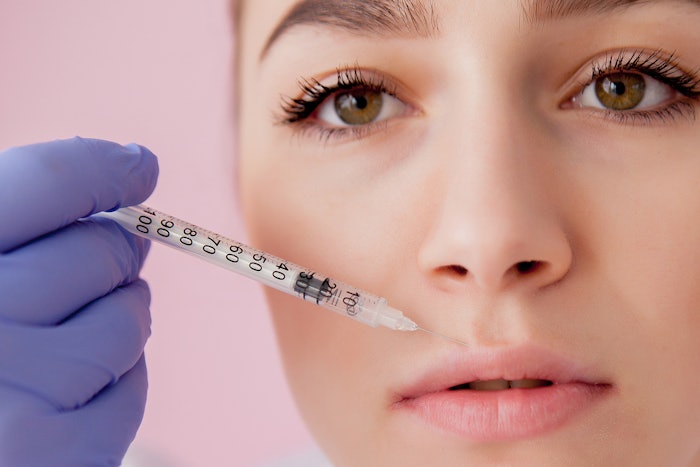
Silk Medical Aesthetics has announced the results of a 44-patient feasibility study under an FDA-approved IDE clinical trial for its ABOUT FACE BY NATURE dermal fillers, which combine hyaluronic acid with the company's Activated Silk protein, made by dissolving pure silk in water.
The fillers are designed to be biocompatible, biodegradable and reversible for soft tissue augmentation.
The 13-week clinical trial results demonstrated a favorable safety profile with high patient satisfaction and clinically meaningful improvement in nasolabial fold (NLF) severity. At the end of the trial, no device-related issues were reported, and common injection site responses, such as discoloration, redness, swelling, pain, tenderness, lumps, bruising and itching, occurred at low rates with most resolving on their own within two weeks.
At 13 weeks, 83.7% of patients showed improvement in wrinkle severity, with a mean improvement of 1.0 in the Wrinkle Severity Rating Scale (WSRS), and 100% of clinicians and patients reported improvement on the Global Aesthetic Improvement Scale (GAIS).
The clinical trial will continue to follow patients over the course of the next 12 months.