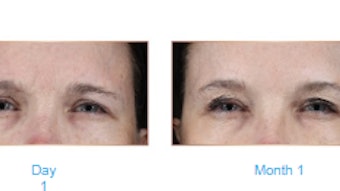
Treatment with a non-cross-linked hyaluronic acid (HA) based soft-tissue filler significantly increased skin firmness and visco-elasticity in the lateral canthal and perioral region in a study published in the Journal of Cosmetic Dermatology (September 2021).
Related: Best Practices When Using Hyaluronic Acid Fillers for Tear Trough Deformities
Researchers Monika Sulovsky, MD et. al, assessed a total of 59 female Caucasian patients with a mean age of 52.6 ± 9.0 in a prospective open-label, multicenter study. The patients received intradermal injections of a soft-tissue filler at baseline, and after three and six weeks. Aesthetic improvement, patient satisfaction, skin hydration, skin firmness and skin elasticity were all assessed at weeks three, six, eight, 12 and 16.
Related: Hyaluronic Acid Injections for Undereye Circles
The lateral canthal skin firmness increased after eight weeks from 0.206 ± 0.07 mm to 0.087 ± 0.08 mm. The perioral skin firmness also increased after eight weeks from 0.205 ± 0.09 mm to 0.116 ± 0.08 mm. While skin hydration did increase after eight weeks, it never reached a point of statistical significance. At weeks eight, 12 and 16 a majority of the patients stated that they were 'satisfied' or 'very satisfied' with the treatment (93.1%, 91.1% and 73.7%, respectively).
Related: FDA Do Not Use Needle-Free Devices for Injection of Dermal Fillers
These findings showed significant increases in the lateral canthal and perioral skin firmness and visco-elasticity following treatment with the non-cross-linked HA filler. The procedure was also shown to be safe and satisfactory for the treated patients. The authors noted that the emergence of oral herpes should be added to the safety profile of intradermally applied HA treatments.