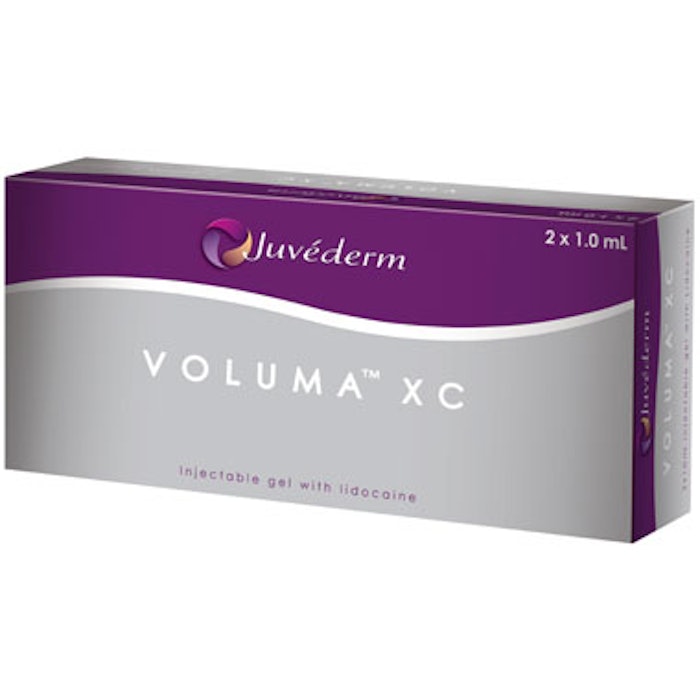
The U.S. Food & Drug Administration (FDA) has approved JUVEDERM VOLUMA XC for the augmentation of the chin region to improve the chin profile in adults over the age of 21. It is the first and only filler to receive FDA approval for chin augmentation. In the U.S. pivotal study, JUVEDERM VOLUMA XC was shown to be safe and effective for augmentation of the chin, with the majority of patients reporting high satisfaction throughout the one-year follow-up.
“When assessing facial anatomy, the chin is a critical component to facial balance, yet it is often overlooked. Due to its distinct gel properties and product profile, JUVEDERM VOLUMA XC is uniquely suitable to address this need,” said Carrie Strom, senior vice president, AbbVie, and president, Global Allergan Aesthetics. “It’s always rewarding when we can offer a safe and effective nonsurgical alternative to patients.”
“Within my practice, I’ve seen a growing demand for patients looking to achieve more balanced symmetry in the face. During facial assessments, most patients don’t realize that chin augmentation is another component to achieving the facial harmony they desire,” said Dee Anna Glaser, MD, dermatologist in St. Louis and clinical trial investigator. “This approval is a truly exciting milestone for providers as it signals another paradigm shift in the way we are able to approach a facial consultation. I am excited to educate patients that we have a safe and predictable nonsurgical treatment option to address their concern.”