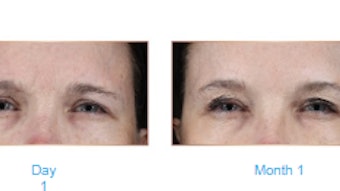
Investigators Joely Kaufman-Janette, MD, et al, established the safety of prabotulinumtoxinA (Jeuveau, Evolus) injections for the treatment of moderate to severe glabellar lines in adult patients in two one-year, multicenter, open-label, repeat-dose, phase II safety studies. The outcomes of the initial study and confirmation study were published in the Aesthetic Surgery Journal (December 2021).
Related: PrabotulinumtoxinA in Adults with Skin of Color: Efficacy and Safety
For the first study, 352 adults with moderate to severe glabellar lines at maximum frown received an initial treatment of 20 U prabotulinumtoxinA (4 U/0.1 mL freeze-dried formulation injected into five target glabellar sites), followed by two additional treatments for a total of three treatments in one year.
Fifty-one patients (14.5%) experienced adverse events (AEs) that were considered possibly study drug-related; 11.1% experienced study drug-related AEs after the initial treatment with the rate of AEs decreasing after each repeat treatment.
Six patients (1.7%) experienced study drug-related AEs of special interest: three eyelid ptosis (0.9%), two speech disorder (0.6%) and one blepharospasm (0.3%).
Related: Reducing the Risks of BTXA-induced Eyelid Ptosis
For the second study, 570 subjects received a median total dose of 60 U over the course of three treatments. Sixty-one patients (10.7%) experienced adverse events (AEs) assessed as possibly study drug related; 6.5% experienced study drug-related AEs after the initial treatment.
As with the first study, the rate of AEs decreased with repeat treatment. Eight patients (1.4%) experienced study drug-related AEs of special interest: five eyelid ptosis (0.9%), three eyebrow ptosis (0.5%), one blepharospasm (0.2%) and one blurred vision (0.2%).
The authors concluded that the safety of repeat treatment of 20 U of prabotulinumtoxinA for moderate to severe glabellar lines was established in the first study and confirmed in the second phase II study.