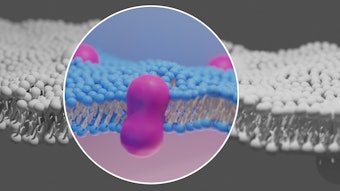
Journey Medical Corporation announced that it has fully enrolled and randomized all of the patients in its Phase 3 clinical program to assess the safety, efficacy and tolerability of DFD-29 (Minocycline Modified Release Capsules 40 mg) for the treatment of papulopustular rosacea (PPR).
DFD-29 vs. Oracea in Rosacea
The clinical trials are part of a collaboration with Dr. Reddy's Laboratories for the ongoing development and commercialization of the DFD-29 program.
“We are thrilled to have completed enrollment in our two DFD-29 Phase 3 clinical trials and look forward to announcing topline data in the first half of 2023, with a New Drug Application (“NDA”) filing subsequently expected in the second half of 2023," said Claude Maraoui, Co-Founder, President and Chief Executive Officer of Journey Medical. "Data from the Phase 2 multicenter clinical trial demonstrated that DFD-29 achieved nearly double the efficacy compared to doxycycline capsules 40 mg on reducing total inflammatory lesions and Investigator’s Global Assessment (“IGA”) treatment success, suggesting the potential of DFD-29 as a more effective treatment option for rosacea."
Related: Journey Medical Acquires Topical Minocycline Products
DFD-29 Phase 3 Clinical Trial
The DFD-29 Phase 3 clinical trial program consists of two multicenter, randomized, double-blind, parallel-group, active comparator and placebo-controlled clinical trials, MVOR-01 and MVOR-2 ((Minocycline versus Oracea in Rosacea), that are expected to support an NDA submission in the U.S. and potentially a Marketing Authorization Application in Europe.
The combined enrollment target of 640 total adult patients with moderate to severe PPR was achieved in the trials; one trial was enrolling patients in the U.S., and the other was enrolling patients in both the U.S. and Europe. The MVOR-01 and MVOR-02 clinical trials are randomized in a 3:3:2 ratio to DFD-29 (Minocycline Hydrochloride Modified Release Capsules, 40 mg), Oracea (Doxycycline capsules 40 mg) or placebo once daily for 16 weeks.
The primary objective is to evaluate the safety, efficacy and tolerability of DFD-29 compared to placebo for the treatment of PPR. The secondary objective is to evaluate the safety, efficacy and tolerability of DFD-29 compared to Oracea (Doxycycline capsules 40 mg). To date, no major safety issues have been reported, and no drug-related serious adverse events have been observed.
Greater Treatment Efficacy
Data published in The Journal of Clinical and Aesthetic Dermatology determined DFD-29 40 mg demonstrated significantly greater efficacy than doxycycline 40 mg, placebo and DFD-29 20 mg for the treatment of PPR in a Phase 2 clinical study. DFD-29 40 mg demonstrated statistical significance compared to both placebo and doxycycline 40 mg on both co-primary endpoints—proportion of subjects with IGA treatment success (grade 0 or 1 with at least a two-grade reduction from baseline at week 16) and total inflammatory lesion count reduction from baseline to week 16.